Mastery Check #5 - Phase Changes and Density
|
Temperature and Particle Motion
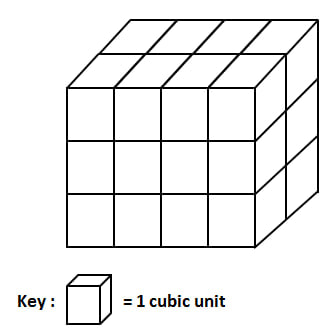
The first key idea that you need to understand is the relationship between the temperature of a substance and the motion of the particles in the substance. Even in a solid, the particles of matter are moving. The hotter that substance becomes, the faster the particles in the substance will be moving. Watch the video to the right to see how this works. Before we move on further, you need to understand volume and density. Volume is the the amount of space an object occupies. For example, to calculate the volume of the cube below, you need to figure out how many cubic units are inside of it. Use Picture #1 to calculate the volume of the object below. Length x width x height 4 x 2 x 3 = 24 cubic unit |
|
|
Density is how much mass occupies the volume of matter. Density is expressed as mass/volume. To calculate density, you will take the mass of an object divided by the volume of an object. If the cube above has a mass of 48 grams, let's calculate it's density.
Mass ÷ volume = density 48 grams ÷ 24 cubic units = 2 grams/cubic unit (we would say 2 grams per cubic unit) Make sure you know how to calculate density!!! Below are the relationships between temperature and volume. Warmer temperatures = Faster movement of particles Faster movement of particles = Increased volume Colder Temperatures = Slower movement of particles Slower movement of particles = Decreased volume When you heat up a substance, the molecules will move faster and as such, there will be more empty space in between the molecules of matter. This will cause the substance to increase in volume. When you cool a substance, the molecules movement will slow and there will be less empty space in between the particles of matter. This will cause the volume of the substance to decrease. Look at Picture #2 to solidify this idea. Then watch the video to the right to see this concept demonstrated. |
|
|
Water as the exception to the rule
As you just learned, the colder you get something, the less space it occupies and the greater it's density. This means that solid iron would sink in liquid iron because it has a higher density. This rule applies to almost all substances, but water is an exception to this rule. Water has some unique properties that cause it to break this rule of density. When water freezes, it actually expands and becomes less dense than liquid water (that is why ice floats in your glass of water, or why ice bergs float in the ocean and boats crash into them). You need to understand that this is not the norm and that most substances are more dense as a solid than as a liquid. If you want to learn more about this, you can watch the video to the right. |
|
|
Phase Changes:
Now that you understand the relationship between temperature, volume, and density, let's see what happens as substances go through phase changes. As you can see in Picture #1, solids are most dense, liquids are in the middle, and gases are the least dense states for most substances. Thought question: How could you change the density of the solid in Picture #1? (hint, look at the arrows between the beakers) As you add or take away heat energy from a substance, you can cause it to change phases. In order to change a solid piece of iron to a liquid, you need to add heat. If you then heat up that liquid iron even more, you can cause it to change phase again into a gas. Then by cooling the substances (removing heat energy), you can cause them to change phases back into a liquid or a solid if you remove a lot of heat. |
|