8th Grade Test Study Guide
2. How our theory of atomic structure ever changed? If so, why?
Our theory changes every time we make new discoveries about the atom. We used to think that atoms were the smallest thing, but then we discovered that there were smaller charged parts and that the positive parts were concentrated in the nucleus.
3. How many protons, neutrons, and electrons does phosphorus have?
According to the periodic table, phosphorus has 15 protons, 15 electrons and 16 neutrons
4. How man protons, neutrons, and electrons does arsenic have?
Protons = 33, electrons = 33, neutrons = 42
5. How many protons, neutrons, and electrons does fluorine have?
Protons = 9, electrons = 9, neutrons = 10
6. How many protons, neutrons, and electrons does copper have?
Protons = 29, electrons = 29, neutrons = 35
7. How many protons, neutrons, and electrons does boron have?
Protons = 5, electrons = 5, neutrons = 6
8. How do we decide which substances to use for a particular function?
We use them according to the properties that they have
9. Why do we use rubber for tires?
Rubber is durable, it can withstand heat, it is flexible
10. Why do we use diamonds for drill bits?
It is very hard
11. Why do we use helium and not hydrogen in balloons?
Hydrogen is flammable and helium is not.
12. How could you change a solid to a gas?
Add energy to it
13. How could you change a gas to a liquid?
Take energy away from it
14. How could you change a liquid to a solid?
Take energy away from it
15. How could you change a liquid to a gas?
Add energy to it
16. How are temperature and density related?
When you heat something up, it expands and gets less dense. When you cool it down it gets more dense.
17. If you heat up a balloon, what happens to its density?
It becomes less dense
18. What are 4 phenomena that could indicate that a chemical reaction has taken place?
Energy released as heat or light
Bubbling or fizzing
A color change
A precipitate forms - mixing two liquids and getting a solid
19. What natural resource does plastic come from?
Crude oil or petroleum
20. What are the pros and cons of using plastic for cups instead of glass?
Pros: Cheaper, doesn't break as easily, flexible
Cons: Bad for the environment, don't look as good
21. Draw what a solid, liquid, and gas would look like in a beaker.
Our theory changes every time we make new discoveries about the atom. We used to think that atoms were the smallest thing, but then we discovered that there were smaller charged parts and that the positive parts were concentrated in the nucleus.
3. How many protons, neutrons, and electrons does phosphorus have?
According to the periodic table, phosphorus has 15 protons, 15 electrons and 16 neutrons
4. How man protons, neutrons, and electrons does arsenic have?
Protons = 33, electrons = 33, neutrons = 42
5. How many protons, neutrons, and electrons does fluorine have?
Protons = 9, electrons = 9, neutrons = 10
6. How many protons, neutrons, and electrons does copper have?
Protons = 29, electrons = 29, neutrons = 35
7. How many protons, neutrons, and electrons does boron have?
Protons = 5, electrons = 5, neutrons = 6
8. How do we decide which substances to use for a particular function?
We use them according to the properties that they have
9. Why do we use rubber for tires?
Rubber is durable, it can withstand heat, it is flexible
10. Why do we use diamonds for drill bits?
It is very hard
11. Why do we use helium and not hydrogen in balloons?
Hydrogen is flammable and helium is not.
12. How could you change a solid to a gas?
Add energy to it
13. How could you change a gas to a liquid?
Take energy away from it
14. How could you change a liquid to a solid?
Take energy away from it
15. How could you change a liquid to a gas?
Add energy to it
16. How are temperature and density related?
When you heat something up, it expands and gets less dense. When you cool it down it gets more dense.
17. If you heat up a balloon, what happens to its density?
It becomes less dense
18. What are 4 phenomena that could indicate that a chemical reaction has taken place?
Energy released as heat or light
Bubbling or fizzing
A color change
A precipitate forms - mixing two liquids and getting a solid
19. What natural resource does plastic come from?
Crude oil or petroleum
20. What are the pros and cons of using plastic for cups instead of glass?
Pros: Cheaper, doesn't break as easily, flexible
Cons: Bad for the environment, don't look as good
21. Draw what a solid, liquid, and gas would look like in a beaker.
22. Which state of matter is generally the most dense and why?
A solid because the particles are moving slower and are closer together.
23. 9,756 grams of a substance occupy 35,000 cubic centimeters of space. Calculate the density of the substance.
9,756 divided by 35,000 = 0.2787 g/cubic centimeters
24. What advantage does a synthetic sweetener like splenda have over a natural sweetener like sugar?
It has no calories
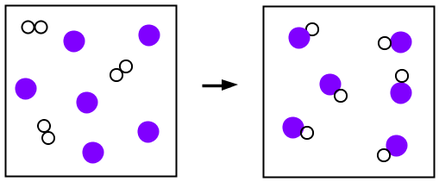
25. Draw a model of a chemical reaction that obeys the law of conservation of mass.
In the picture below, there are the same number of white and purple atoms on each side, even though they have rearranged into new molecules.
A solid because the particles are moving slower and are closer together.
23. 9,756 grams of a substance occupy 35,000 cubic centimeters of space. Calculate the density of the substance.
9,756 divided by 35,000 = 0.2787 g/cubic centimeters
24. What advantage does a synthetic sweetener like splenda have over a natural sweetener like sugar?
It has no calories
25. Draw a model of a chemical reaction that obeys the law of conservation of mass.
In the picture below, there are the same number of white and purple atoms on each side, even though they have rearranged into new molecules.
26. If you start out with 100 kilograms of wood and burn it, the ashes only weigh 1.5 kilograms. Where did the other 98.5 kilograms go?
It turned into gases that escaped into the atmosphere
27. Are phase changes chemical changes? Why?
They are not because they don't produce a new substance.
It turned into gases that escaped into the atmosphere
27. Are phase changes chemical changes? Why?
They are not because they don't produce a new substance.