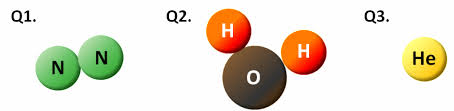Chemistry Mastery Check #1 - Scale and Proportion of Atoms
|
Reading the Periodic Table
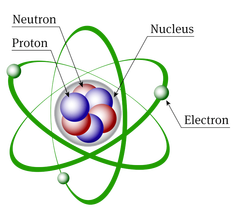
Picture #1 shows the periodic table tile of Argon. There are several things you will need to be able to do when looking at the periodic table. The tiles on the periodic table give you all of the information about the subatomic parts of that atom. There are 3 subatomic particles that we are concerned with in an atom; the protons, neutrons, and electrons.
Below is a summary of how to determine the numbers of these subatomic particles. The video to the right will also help you as you learn how to determine the numbers of subatomic particles in each atom.
|
|
|
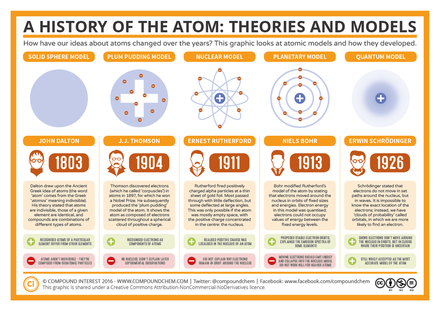
Our Evolving Understanding of Atoms
|
|
The 3 Big Advances in Atomic Theory:
Could our current theory of atomic structure change? You bet! If we find compelling evidence that the structure of atoms is different than how we view them now, our theories could change. But that evidence would have to be reinforced by scientific experimentation. |
|
|
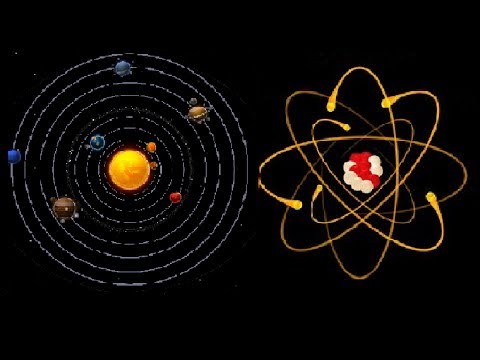
Scale and Proportion of Atoms
Our solar system is a fair representation of atomic structure. If we compare the nucleus of an atom to the sun and the planets to electrons, we may better understand the size and scale of an atom. There are a few key similarities in this comparison:
|
|
Atoms vs. Molecules
A molecule is a combination of 2 or more atoms. For example, water is a molecule that is made up of 2 hydrogen atoms and 1 oxygen atom. Picture #1 shows how this works. The oxygen you are breathing is actually 2 oxygen atoms bonded together so you are breathing oxygen molecules (see Picture #2). Test yourself by determining whether the 3 examples below are atoms or molecules. |
|