Chemistry Quiz Study Guide
1. What would a scale model of an atom look like? Would you be able to draw one on a single piece of paper?
The atoms is mostly empty space. This means that the nucleus is really small compared to the rest of the atom and most of the mass of the atom is contained in the nucleus. If you we to expand the nucleus so that it were the size of a marble, then the electrons would be miles away. This means that you could never represent it on a single piece of paper.
2. What are the three major discoveries that led to our current model of the atom.
The first discovery was that the atom is made of smaller, charged parts.
The second discovery was that most of the mass was in the center of the atom and it was positively charged. They called this center the nucleus.
The third discovery was that electrons are orbiting in specific orbitals around the nucleus, much like planets around the sun.
If we were to make more discoveries about the atom, we would change our model to represent those new discoveries.
3. What properties make diamonds good for jewelry?
They are shiny, hard, don't react chemically easily, and have a high melting point
4. Use the picture to figure out how many protons, neutrons and electrons an atom of copper has.
The atoms is mostly empty space. This means that the nucleus is really small compared to the rest of the atom and most of the mass of the atom is contained in the nucleus. If you we to expand the nucleus so that it were the size of a marble, then the electrons would be miles away. This means that you could never represent it on a single piece of paper.
2. What are the three major discoveries that led to our current model of the atom.
The first discovery was that the atom is made of smaller, charged parts.
The second discovery was that most of the mass was in the center of the atom and it was positively charged. They called this center the nucleus.
The third discovery was that electrons are orbiting in specific orbitals around the nucleus, much like planets around the sun.
If we were to make more discoveries about the atom, we would change our model to represent those new discoveries.
3. What properties make diamonds good for jewelry?
They are shiny, hard, don't react chemically easily, and have a high melting point
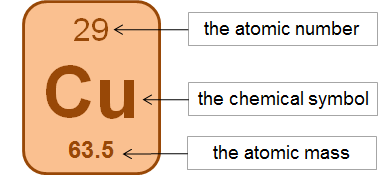
4. Use the picture to figure out how many protons, neutrons and electrons an atom of copper has.
The atomic number is the number of protons. The number of protons = the number of electrons in a neutral atom. The atomic mass is protons and neutrons added together (remember to round to the nearest whole number). There would be 29 protons, 29 electrons and 35 neutrons in a copper (Cu) atoms.
6. Look at the substances below. What are they used for and what property makes them useful for that purpose?
7. What are the 4 signs of a chemical change? Give an example of each.
- Gasoline is used for fuel in cars because it is flammable, it contains a lot of energy, and it is a liquid.
- Iron is used to make steel because it is strong and malleable.
- Rubber is used for kickballs because it is soft, elastic, and doesn't allow air to pass through it.
- Paper is good for books because it is light, cheap, and can absorb ink.
- Wood is good for a baseball bat because it is hard, can be easily shaped, and is light.
- Gold is good for wedding rings because it doesn't rust, it is shiny, and it is malleable
- Glass is good for windows because it is see through, hard, doesn't allow air or water to pass through it
7. What are the 4 signs of a chemical change? Give an example of each.
- Color change - cooking an egg (the clear liquid inside turns white, we added dry ice to blue liquid and it turned yellow, we added dry ice to the pink liquid and it went clear
- Energy released - burning anything
- Bubbling or fizzing - baking soda and vinegar - adding baking soda to vinegar makes it fizz and bubble, zinc and hydrochloric acid - when combined, they get really hot and produce hydrogen gas that bubbles
- Forming a precipitate - we added two clear liquids together and a brown solid substance formed. On the video, two clear liquids were added and they formed a solid orange substance