Mastery Check #3 - Energy Transfers
|
The law of the conservation of energy: This law states that energy cannot be created or destroyed; it can only change forms (kinetic to heat, etc.).
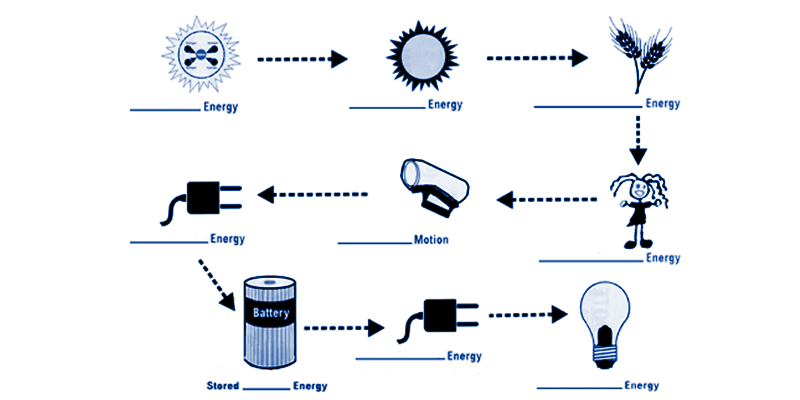
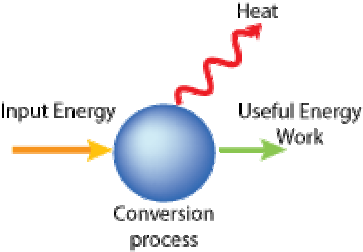
According to the law of the conservation of energy, if an object has any type of energy, that same amount of energy will remain in the system but it can change forms. For example, if a car has 10,000 joules of kinetic energy and the car then comes to a stop, what happened to the energy? You may be tempted to say that it disappeared but that would be incorrect. All 10,000 joules of that energy had to have changed into some other type of energy. In the case of the car, the mechanical energy was transformed into heat in your braking system. (see Picture #1) Complete the picture to the right to help you understand energy conversions. |
|
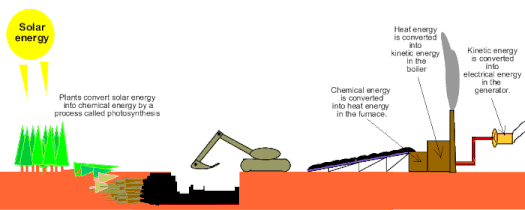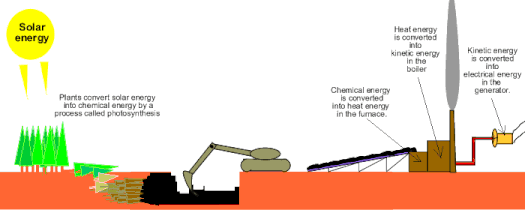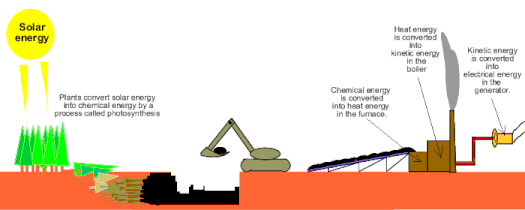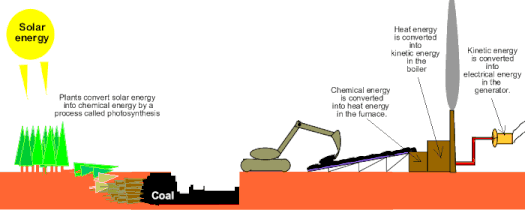
Power Plants: Let's assume that we have a coal burning power plant that creates electricity in Alpine. When you want to listen to music in your headphones, there are a few energy conversions that have to happen in order to do that. First, chemical energy is converted to electrical energy in the power plant and then that electrical energy is converted to sound (mechanical energy) in your headphones.
|
|
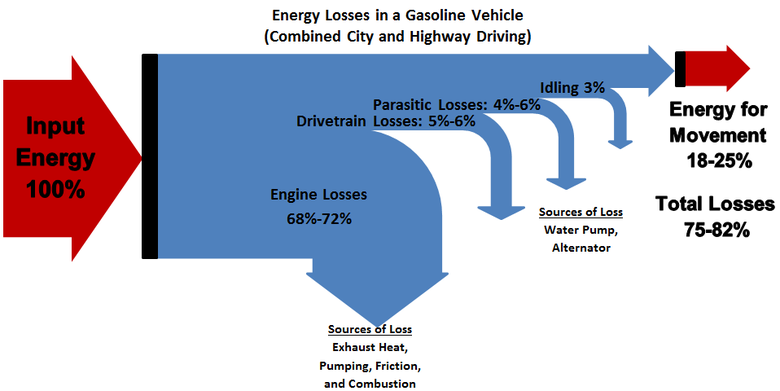
Energy is often lost to heat: The energy of a system is often lost as heat. For example, a roller coaster will eventually come to a stop if it is on flat track but where did all of it's energy go? It was lost to heat due to the friction of the rolling wheels. So it is with many energy conversions. Another example of this is your car's engine. Gasoline has chemical energy which is turned into mechanical energy inside the engine, but only a portion of the chemical energy is converted to mechanical energy. The rest is converted to heat which your car then dissipates in the radiator. (see Picture #3) Additionally, our bodies turn the chemical potential energy in our food into energy we use for our needs such as mechanical energy to be able to move, electrical energy that drives the signals in our nervous system and finally heat energy to keep us warm,
|
Collisions: In the case of collisions, kinetic energy can be transferred to or from objects. For example, remember our kinetic energy demonstrating tables (pool tables), what happens to the kinetic energy of the balls when they collide with other balls? The same laws that apply in pool apply in the real world as well. When you tried to play pool with a ping pong ball, it didn't work because the ping pong ball didn't have as much kinetic energy due to its lower mass and as such it had very little effect on the kinetic energy of other objects on the table. Now imagine you have a light car that hits a heavier car and predict the results by comparing it to the model of the pool table. What would happen if a semi truck that has lots of mass was to rear end a passenger car with relatively low mass? Use this online collision simulator to help you understand this concept. Also, examine the picture below to help you understand this concept. The picture below shows how the calculations for this work, while you don't need to know how to do these calculations, it is helpful to see how the numbers work out in a situation like this. Spend a few minutes analyzing this situation to see how it works.