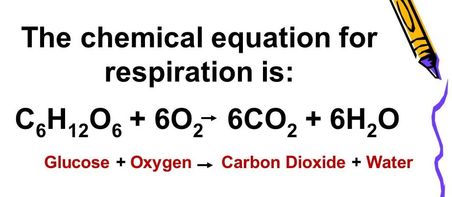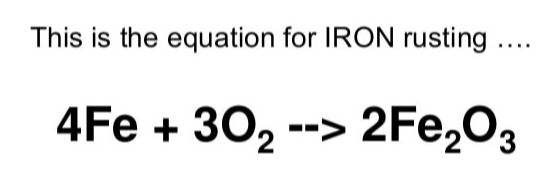Chemistry Quiz #1 Study Guide
1. Using the picture below, determine each of the following:
- The number of protons in the nucleus of Beryllium. Protons = 4 (the number above the symbol).
- The number of neutrons in the nucleus of Beryllium. Neutrons = 5 (round 9.012182 to 9 and then subtract 9-4 = 5)
- The number of electrons in a neutral atom of Beryllium. Electrons = 4 (if it's a neutral atom then protons = electrons)
- The number of valence electron Beryllium has. Valence electrons = 2 (because it's in the second column)
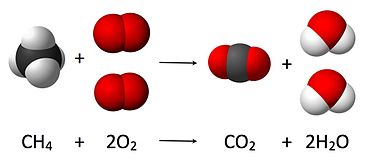
2. What is the chemical equation for combustion of methane (CH4)?
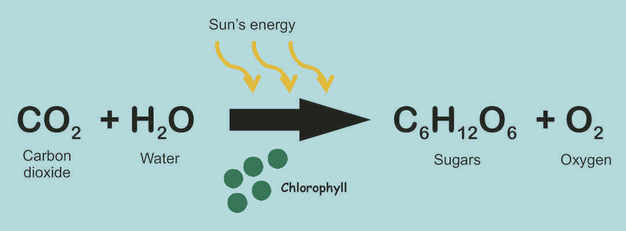
3. What is the chemical equation for photosynthesis?
4. What is the chemical equation for cellular respiration?
5. What is the chemical equation for rusting?
6. Why do atoms make chemical bonds?
The bond with other atoms so they can get a full outer layer of electrons
7. Draw a model of an atom and label the protons, neutrons and electrons.
The bond with other atoms so they can get a full outer layer of electrons
7. Draw a model of an atom and label the protons, neutrons and electrons.
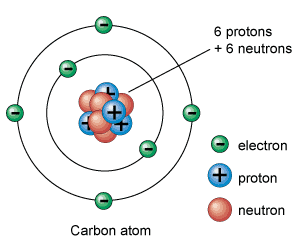
8. What is mass number and what is the mass number of the atom pictured above?
It is the number of protons and neutrons. There are 12 protons and neutrons in the picture above.
9. What is atomic number and what is the atomic number of the atom pictured above?
It is the number of protons. There are 6 protons in the picture above.
10. What do all of the elements in the same column have in common?
They have the same number of valence electrons (electrons in their outermost energy level)
It is the number of protons and neutrons. There are 12 protons and neutrons in the picture above.
9. What is atomic number and what is the atomic number of the atom pictured above?
It is the number of protons. There are 6 protons in the picture above.
10. What do all of the elements in the same column have in common?
They have the same number of valence electrons (electrons in their outermost energy level)
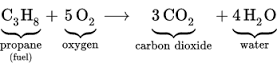
11. How many molecules of carbon dioxide are in the equation above?
There are 3
12. How many total oxygen atoms are there on the right side of the equation above?
There are 10 total (6 from the CO2 and 4 from the H2O)
13. How many atoms are on the left side of the equation above compared to the right side?
There are the same number of atoms on each side. Atoms never disappear or come from nowhere.
14. What are the products and reactants in the equation above?
Products = CO2 and H2O
Reactants = C3H8 and O2
15. What is a chemical change?
A change that produces new molecules. Molecules are rearranged to make new molecules.
16. What is a physical change?
When a substance changes somehow but it stays the same substance (new molecules are NOT made)
17. What are some common chemical changes?
Respiration
Burning something
Rusting or corroding metal
Photosynthesis
18. What are some common physical changes?
Dissolving substances
Phase changes (freezing, melting, etc.)
Heating or cooling something
Changing density or volume of a substance
19. What are the 4 signs of a chemical change? (note: you'll have to write these out on your quiz)
Color change
Bubbling or fizzing
Heat or light released
Precipitate forms
There are 3
12. How many total oxygen atoms are there on the right side of the equation above?
There are 10 total (6 from the CO2 and 4 from the H2O)
13. How many atoms are on the left side of the equation above compared to the right side?
There are the same number of atoms on each side. Atoms never disappear or come from nowhere.
14. What are the products and reactants in the equation above?
Products = CO2 and H2O
Reactants = C3H8 and O2
15. What is a chemical change?
A change that produces new molecules. Molecules are rearranged to make new molecules.
16. What is a physical change?
When a substance changes somehow but it stays the same substance (new molecules are NOT made)
17. What are some common chemical changes?
Respiration
Burning something
Rusting or corroding metal
Photosynthesis
18. What are some common physical changes?
Dissolving substances
Phase changes (freezing, melting, etc.)
Heating or cooling something
Changing density or volume of a substance
19. What are the 4 signs of a chemical change? (note: you'll have to write these out on your quiz)
Color change
Bubbling or fizzing
Heat or light released
Precipitate forms